|
|
|
Drug Development
Search |
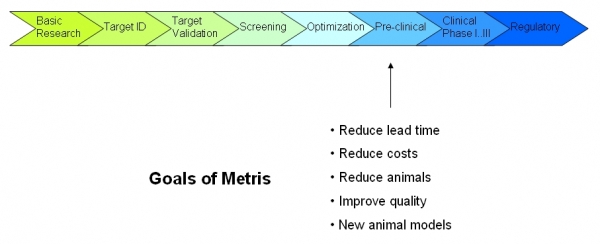
Drug DevelopmentDrug development is defined in many pharmaceutical companies as the process of taking a new chemical lead through the stages necessary to allow it to be tested in human clinical trials, although a broader definition would encompass the entire process of drug discovery and clinical testing of novel drug candidates (Wikipedia, Free Encyclopedia). The process of developping a new medicine is given below:
|
Additional information
Upcoming conferences IASP 2024
Latest News Metris introduces USV Classifiication software ...
|
| Site map | Privacy Statement | Animal Welfare | Feedback |